This article is not intended to be an all-inclusive guide to time tested underwriting guidelines but rather is designed to shed some light on an often misunderstood concept in the surety industry. What is my bonding capacity? This may be one of the most frequently asked questions by contractors and probably the most difficult to answer on an on-going basis. First, the bond length elongates because of the physical size of the halogen atom, and second, outer-shell electrons are. BondĪs we go down the series, two things happen. The F-H bond is quite strong for a single bond because it only has one full shell of eight electrons (n = 2), which are all (including the single shared electron of the hydrogen atom) held tightly by the proximity of the fluorine nucleus. In the series below, compare strengths of the bonds between fluorine, the most electronegative atom, and H, F, Cl, Br and I. Here the bond strengths diminish with increasing bond length due to the size of the halogen atom. In the carbon-halogen series below, bond strengths and lengths are compared for C-F, C-Cl, C-Br and C-I bonds. Rerrangements in the bonding geometry of CH 4 and the resulting molecule after each successive removal account for the small differences in bond strength. Notice in the table above that removing the last hydrogen takes less energy, and that removing the second takes the most. While all C-H bonds of methane (CH 4) are and ought to be equivalent, they are not equivalent in this series showing the energy needed to remove the next H atom. Here are strengths (in KJ/mol) and lengths in Ångstroms (Å) (1 Å = 1 × 10 -10m) of single, doulbe and triple bonds of between carbon atoms: Bond type Mostly, the stronger a covalent bond, the shorter it is - and it's true the other way around. We'll call that bond order in the section below. It's easy to see in that table the general trend of increasing bond strength with number of electron pairs shared. We will look at many more examples of bonding below. It's why it is polar (has distinct negative and positive "ends") and why it forms hydrogen bonds. That's part of what makes water such an interesting and unique substance. In water the oxygen atom shares an electron with each hydrogen, but the bonding electron pairs are strongly drawn toward the oxygen, leaving a mostly-bare proton sticking out. It turns out that the covalent O-H bonds in water have significant ionic character. 
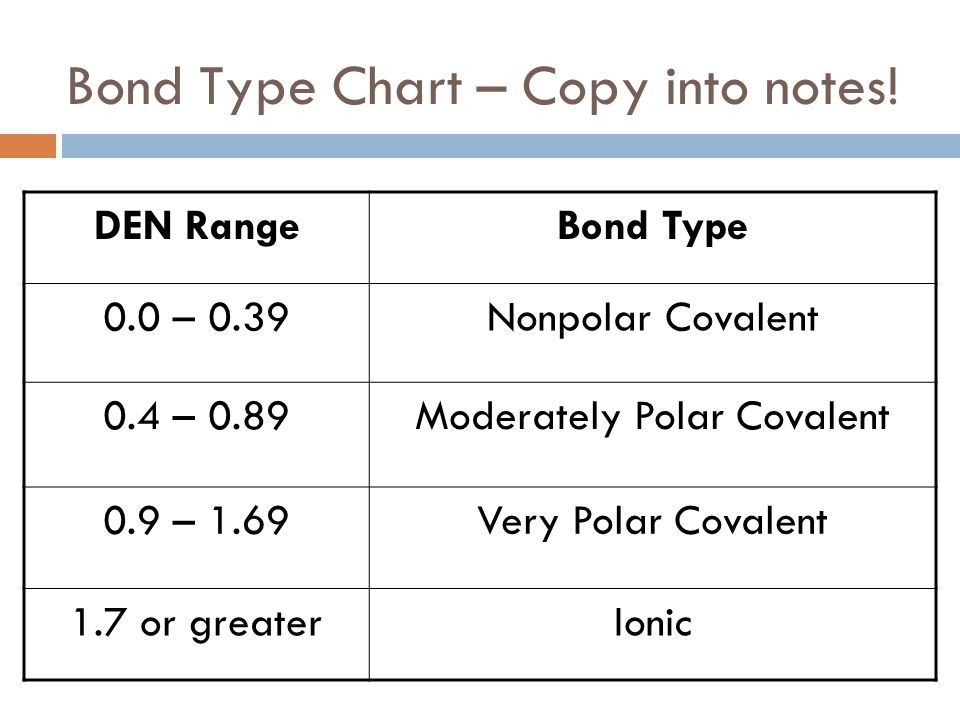
The electronegativities of O and H are 3.44 and 2.20, respectively, for a difference of 1.2. It's a little tricker when the electronegativity falls into that in-between area. The electronegativities of C and H are 2.25 and 2.20, for a small difference of 0.05, so we'd expect these atoms to bond covalently, and they do. Looking at the table below, we see that the electronegativities of sodium (Na) and chlorine (Cl) are 0.93 and 3.16, respectively, for a difference of 2.23, so we'd expect these atoms to bond ionically, which they do. Nevertheless, there are a couple of accepted rules. That's because there are always atom pairs that fall somewhere between ionic and covalent bonding. You've noticed that I haven't said what "large" and "small" actually are. When the difference is small, they'll bond covalently. In general, when the difference between electronegativity of two atoms is large, they will tend to bond ionically. The Pauling electronegativities of most of the elements are given in red in the periodic table below. I won't go into the detail of Pauling's calculation. Electronegativities of the elements can be calculated according to a formula developed by Nobel Prize-winning chemist Linus Pauling (Pauling was the only person to ever have won two Nobels, one in chemistry, the other the Peace Prize for his work to stop the nuclear arms race). One way is to use electronegativity, which we discussed in the periodic trends notes. 
How can we tell how two atoms will bond ahead of time? Atoms bond as groups of ions (usually pairs) or they bond covalently by sharing electrons in bonding orbitals.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
